Table of Contents
Toggle- Before going to the “Gluconeogenesis pathway with enzymes,”
- What is gluconeogenesis?
- Is there any difference between gluconeogenesis and glucogenesis?
- And what is meant by non-carbohydrate or non-glucose sources?
- Usually, glucose is broken down to release energy but here the myth is the opposite.
- Do you know where extra glucose is stored in the body?
- And even when the glucose becomes more after storage in the liver then where does it go for storage?
- And when this extra glucose is released?
- But did you think what will happen if all the liver glycogen is used like in a fasting state?
- But if I am at rest why the body needs glucose?
- But where does gluconeogenesis take place i.e. the organs?
- How does the process start?
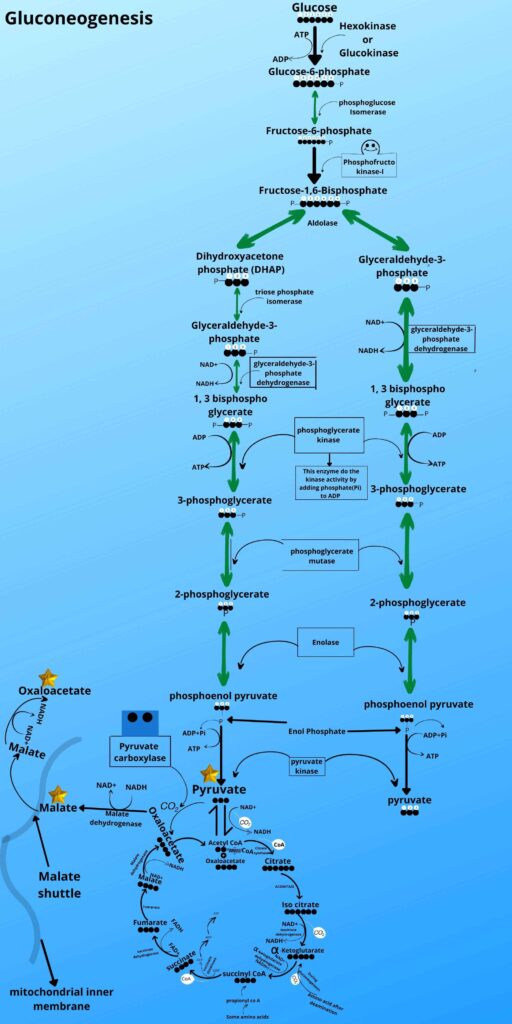
- What are those steps?
- But if there is no glucose how these processes are reversed?
- But just by reversing the Krebs cycle and glycolysis process, we can get new glucose from these raw materials, isn’t it?
- Here I want to tell you about a medical condition.
- But how this enzyme is activated?
- Now let me tell you some of the roles of “protein kinase A”.
- But what is the function of the endoplasmic reticulum here?
- As already discussed gluconeogenesis occurs in the liver and kidney, do you know why?
- Conclusion/summary:
- Source:
Before you go into this article I would highly recommend learning and reading Glycolysis and then after glycolysis, the Krebs cycle, because if you learned these topics 80% of your concept will be clear and 20% of Gluconeogenesis is just here. Before going to “when gluconeogenesis occurs?” let me explain gluconeogenesis a little.
What is gluconeogenesis and how can we define gluconeogenesis?
Just carefully look at the name you will get the meaning.
Gluco = glucose
Neo = new
Genesis = synthesis

So Gluconeogenesis is the synthesis of new glucose from non-carbohydrate or non-glucose sources.
OR
The process of producing glucose from noncarbohydrate sources is called Gluconeogenesis.
Is there any difference between gluconeogenesis and glucogenesis?
Some people search gluconeogenesis as gluconeogenesis vs glucogenesis to know their difference but I would like to tell you that both are the same terms, however, glycogenesis is different which means the synthesis of glycogen from the chaining of glucose.
And what is meant by non-carbohydrate or non-glucose source?
The non-carbohydrate or non-glucose source is the molecules other than carbohydrates like proteins, lipids, etc.
Usually, glucose is broken down to release energy but here the myth is the opposite why?
You, mean why we need GLUCONEOGENESIS and why does gluconeogenesis occur?
Yes.
Let me ask you a question
Do you know where extra glucose is stored in the body?
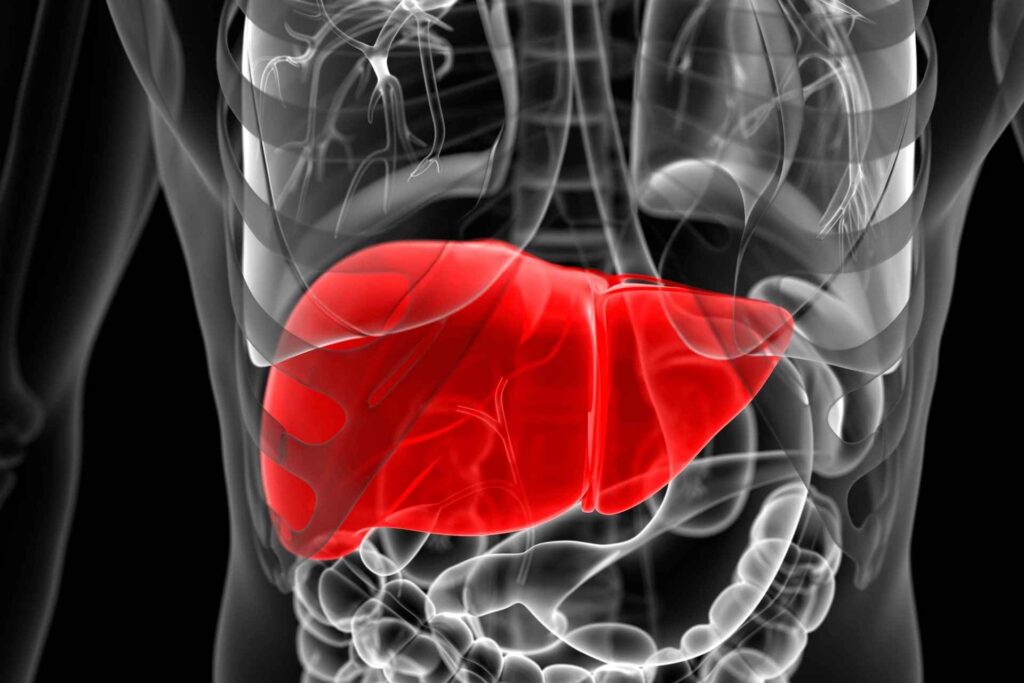
The liver, Liver is the storehouse of glucose where extra glucose is stored in the form of Glycogen.
And even when the glucose becomes more after storage in the liver then where it goes for storage?
Oh yes, that is muscle cells, fat cells (adipocytes).
And when this extra glucose is released?
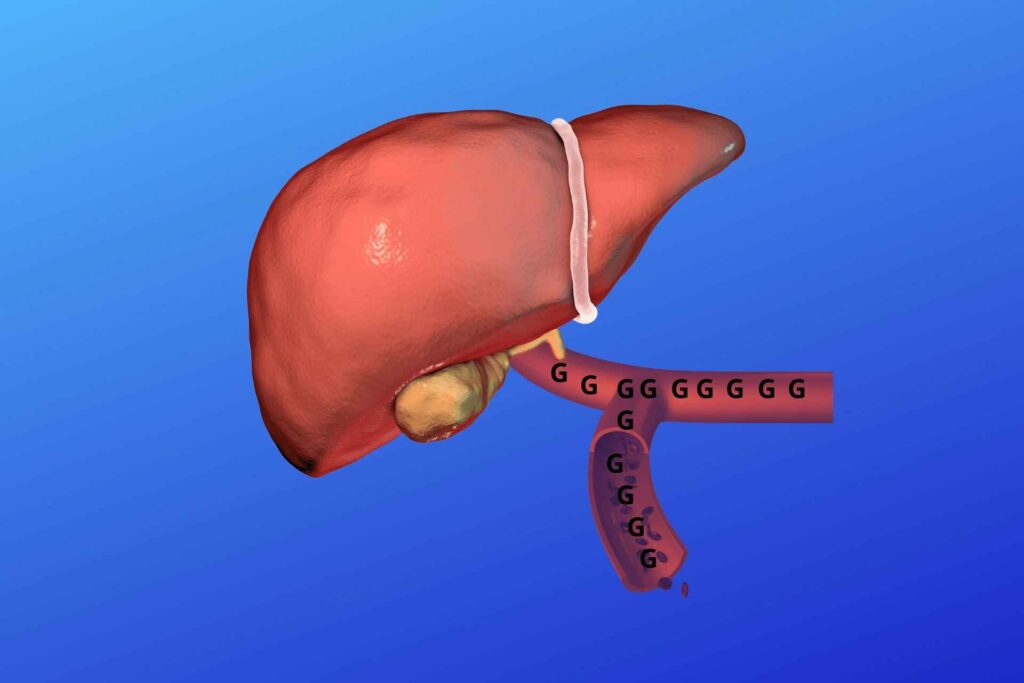
When there is a low blood glucose level then liver glycogen is back converted to glucose and released to blood for their action.
But did you think what will happen if all the liver glycogen is used like in a fasting state?
Not exactly.
That’s why we are discussing “when gluconeogenesis occurs.”
What do you mean?
It means when all your glycogen is used then gluconeogenesis becomes active to synthesize new glucose because in the fasting state there is no supply of glucose from GIT, also the blood glucose level is constantly used.
But if I am at rest why the body needs glucose?
Because
CNS,
RBC,
Renal medulla,
Testes,
Cornea and lens
Require a constant supply of glucose.
But where does gluconeogenesis take place i.e the organs?
One gluconeogenesis organ (location) you know, is the liver where the major gluconeogenesis occurs, but the kidney also does some of the gluconeogenesis while the epithelium of GIT has very little ability to do this.
Okay.
How does the process start?
In the glycolysis and Krebs cycle, the glucose was broken down to release energy but here we have to reverse the process to produce glucose.
So we have just to reverse these processes in the gluconeogenic pathway?
Wait, it is not so just easy to reverse the process.
Why?
Because we have 3 irreversible steps of glycolysis which are one way, it cannot be back directly by the same enzymes.
Then?
The cells have some mechanism to reverse these steps but with different enzymes and different pathways.
What are those steps?
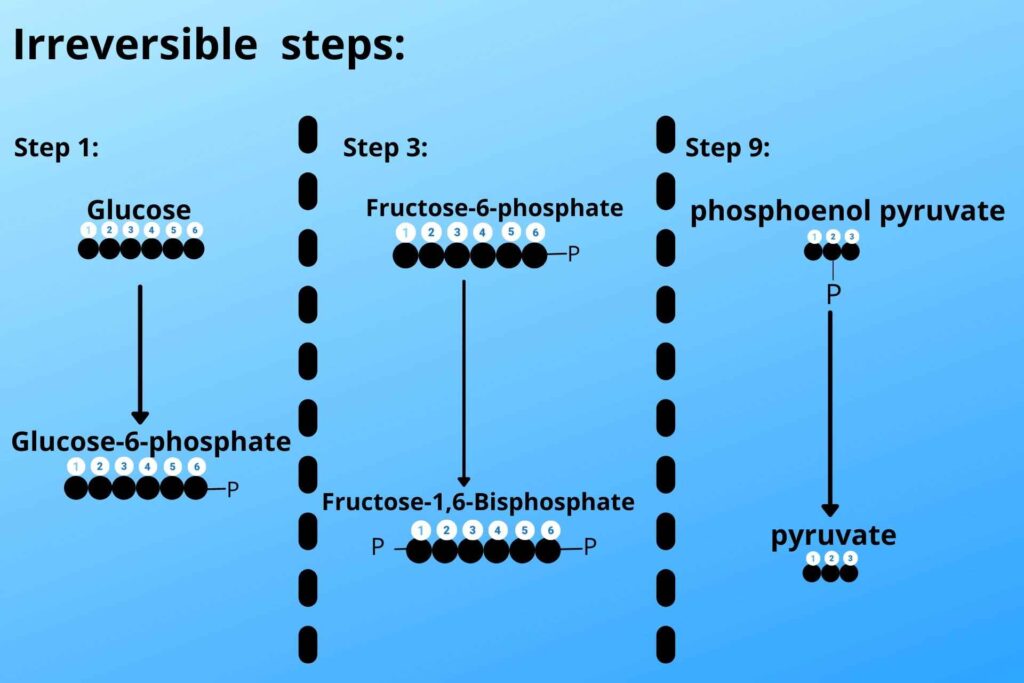
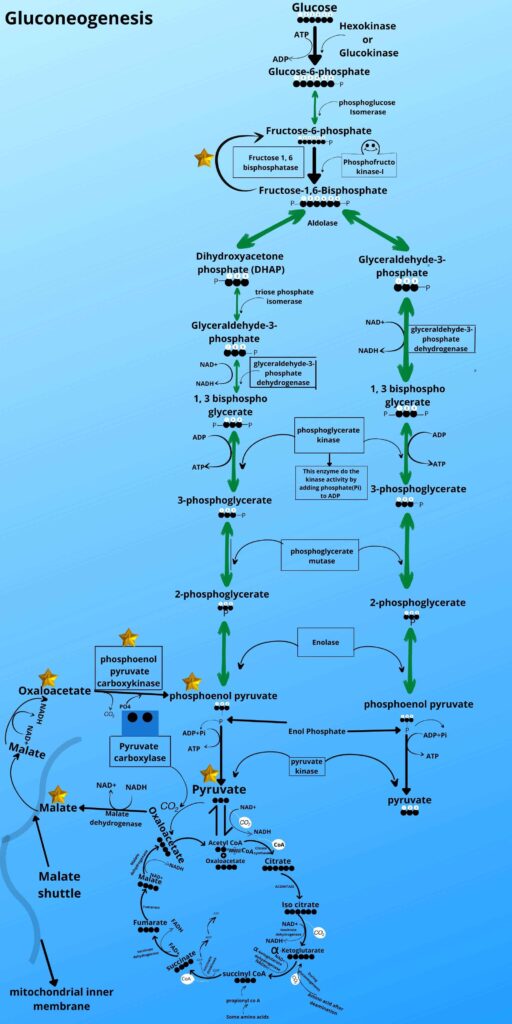
Let me just quickly gives you a very brief review of those steps (in glycolysis) in a picture then I will go with gluconeogenesis that how that step is reversed.
Look at the picture and see steps 1, 3, and step 9 of glycolysis.
But if there is no glucose how these processes are reversed?
Good question. For Gluconeogenesis some raw materials are needed. Glucose is a six-carbon compound so we need some carbon skeleton from which we can make new glucose.
The raw materials for gluconeogenesis are:
Lactate,
Pyruvate,
Alanine,
Triglycerides (adipocytes).
i. Lactate:
From where we can get lactate?
Before going to discuss Lactate you must know that lactate can be converted to pyruvate. An enzyme called Lactate Dehydrogenase (as the name indicates) removes Hydrogen from Lactate and converts it to pyruvate. Hydrogen is used to convert NAD+ into NADH. Pyruvate and lactate are interconvertible molecules.
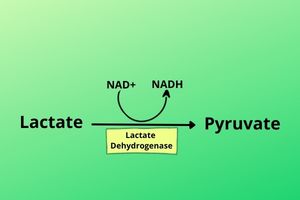
As it is discussed that Lactate can be converted to Pyruvate But from where to get Lactate?
From Muscles and RBCs.
How?
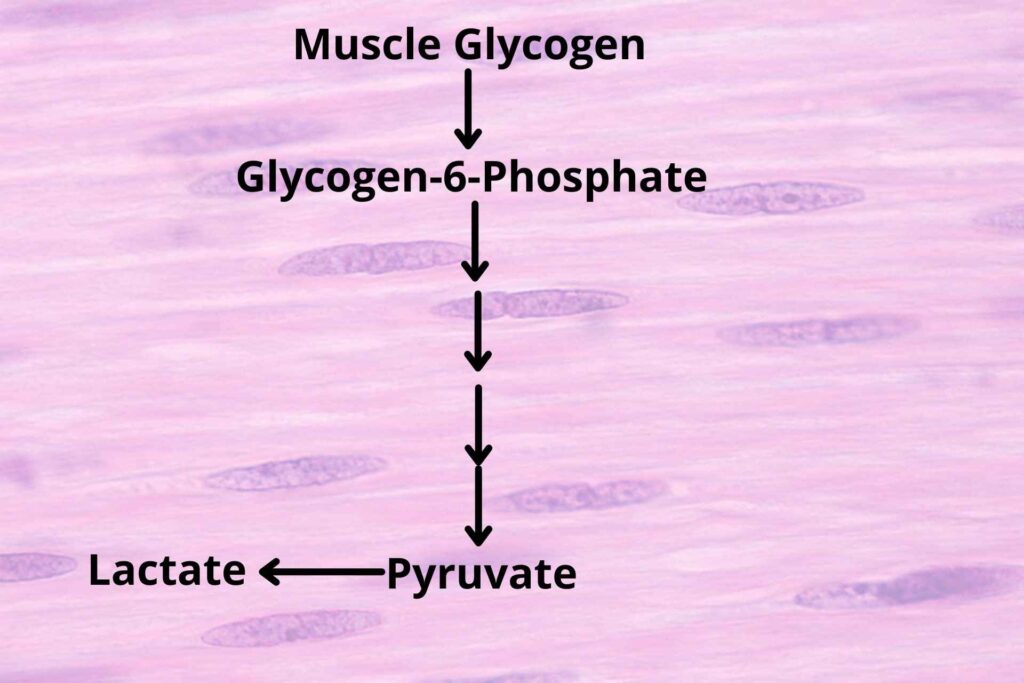
As you know that glucose is also stored in Muscle cells. The muscle cells release Glycogen in the form of Glucose-6-phosphate. But remember that muscle glycogen is not converted to glucose rather it is converted to Glucose-6-phosphate, and also muscle don’t have Glucose phosphatase enzyme which can remove phosphate from Glucose-6-phosphate and convert it to glucose so the muscle cannot directly release glucose to blood rather it converts it into pyruvate after breakdown pathway.
But the product is pyruvate, not lactate?
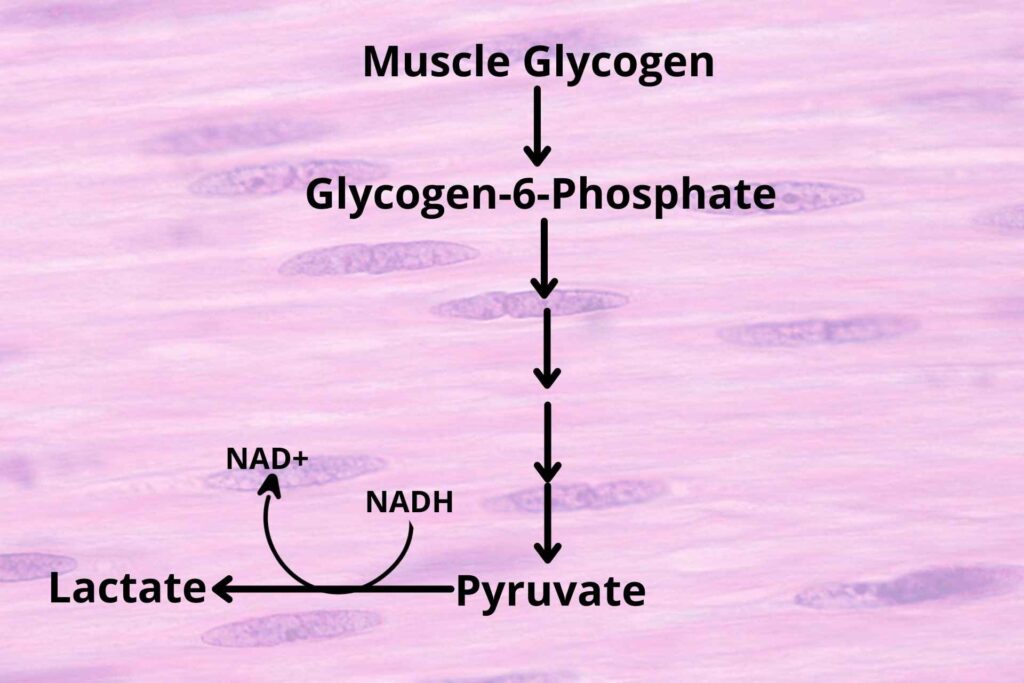
The enzymes which convert pyruvate into Lactate are present in muscle cells so the muscle’s pyruvate is converted into lactate by adding hydrogen to it from NADH and releasing NAD+.
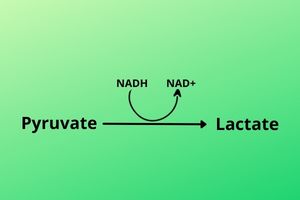
The Lactate is then released into the blood and then move to the liver.
RBC also releases lactate to blood because RBCs don’t have mitochondria so there is anaerobic glycolysis and anaerobic glycolysis produces lactate in their end.
After entry to hepatocytes, it starts the gluconeogenesis pathway steps (will be discussed later).
ii. Pyruvate:
Pyruvate itself sometimes leaks out from muscle cells and enters to gluconeogenesis pathway.
iii. Alanine:
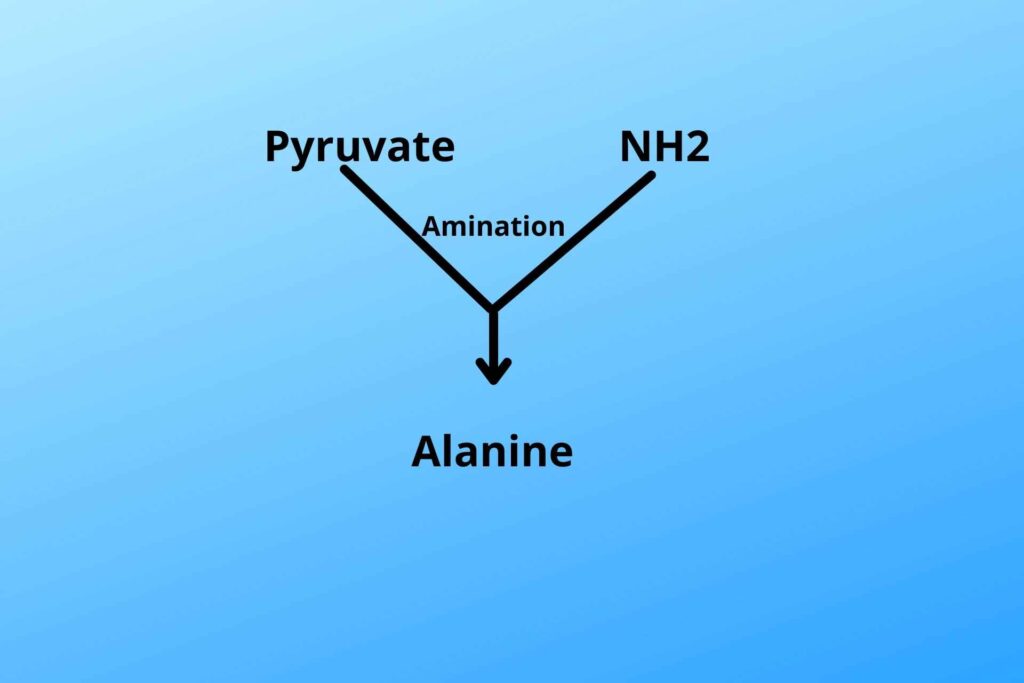
In muscle and amino (NH2) group (Amination) is added to pyruvate which results in the formation of an amino acid called alanine, then alanine is released to blood from where it moves to the liver. In the liver, deamination removes the amino (NH2) from alanine and is converted back to pyruvate in the liver.
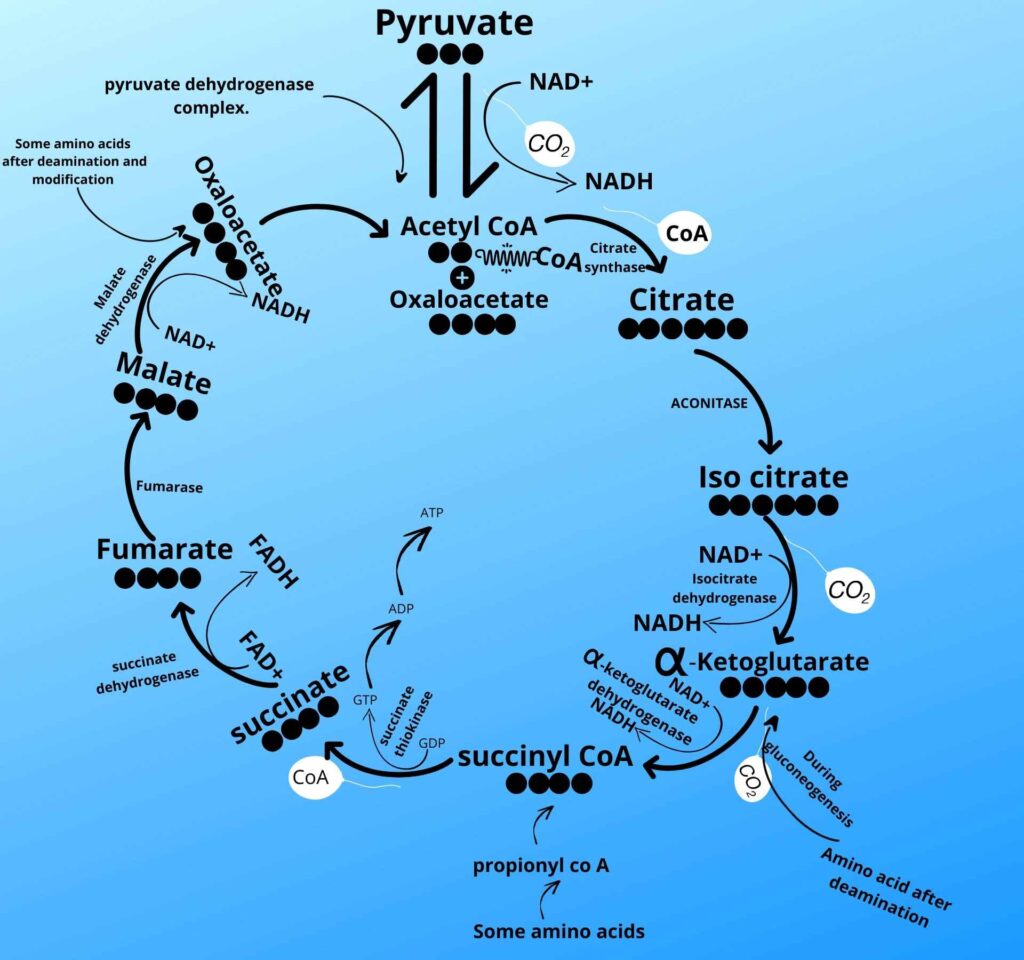
Let me tell you more about amino acids. Most of the amino acids after deamination left with carbon skeleton which is called α-ketoglutaric acid and the α-ketoglutaric acid (carbon skeleton) then reverse the Krebs cycle to synthesize new glucose molecule.
There are some amino acids which is converted to propionyl co A and then propionyl co A is then converted to succinyl co A to reverse the Krebs cycle pathway up to glucose of glycolysis. Propionyl co A is also synthesized from β-oxidation of odd chain fatty acid but this pathway is negligible in humans.
There are some other amino acids which after deamination and further modification are converted to oxaloacetate which again reverse the Krebs cycle to glucose of glycolysis which is known as amino acid gluconeogenesis.
So it means the carbon skeleton of different amino acids after deamination is used to make new glucose from it and those amino acids which provide these carbon skeletons are called gluconeogenic amino acids.
What do you guess from it?
No.
Let me tell you, in prolonged fasting state proteins are catabolized to release amino acids which then participate in gluconeogenesis.
iv. Triglycerides (Adipocytes):
In a fasting state when glucose supply is stopped, the fat cells also called adipocytes to have triglycerides.
Do you know triglycerides?
Not a lot.
Well, triglycerides have three fatty acid molecules with glycerol.
Okay.
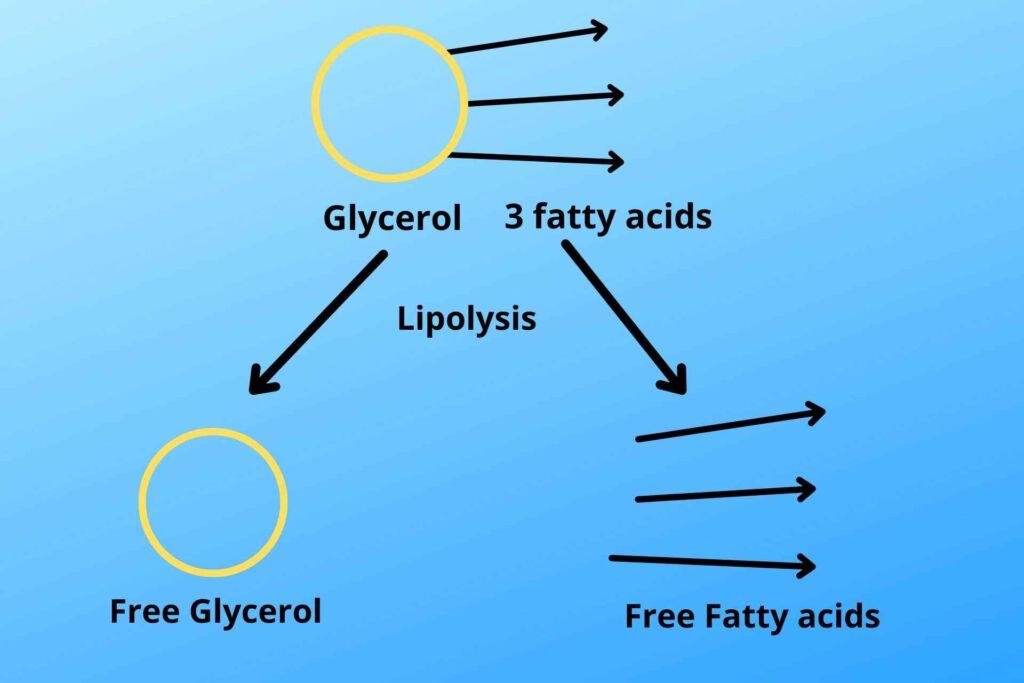
Triglycerides are broken down to free fatty acid and free glycerol from adipocytes by the process of lipolysis. The free glycerol and fatty acids are then released to blood, from where it moves to the liver.
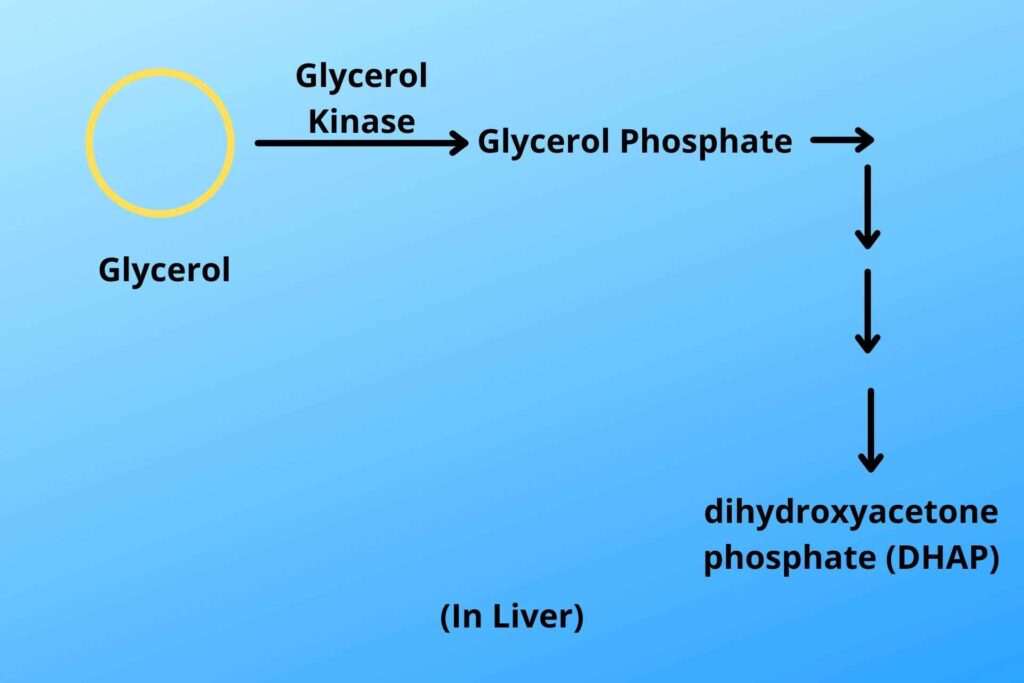
In the liver, an enzyme called Glycerol kinase adds a phosphate to it and converts it to glycerol phosphate and then after multiple conversion steps, it is eventually converted to dihydroxyacetone phosphate which is then reversed to make glucose (will be discussed later)so gluconeogenesis from glycerol is another way to make new glucose.
What about fatty acids?
Fatty acids move to mitochondria where it is converted to Acetyl Co-A and it also activates the enzyme pyruvate carboxylase (will be discussed later).
So we have got the raw materials for gluconeogenesis now we have to convert it to glucose by reversing the process.
But just by reversing the Krebs cycle and glycolysis process, we can get new glucose from these raw materials, isn’t it?
YES, it is not so easy by saying just reversing these processes.
But why?
Because the mechanism of gluconeogenesis has some steps where enzymes can move the reaction forward direction but not in the backward direction, so we have to find some alternative pathway for that steps as shown in the picture.
Which steps are these?
There are three steps which are irreversible i.e
Pyruvate to phosphoenolpyruvate by pyruvate kinase enzyme,
Fructose 1, 6 bisphosphates to Fructose-6-phosphate by phosphofructokinase,
Glucose-6phosphate to Glucose by hexokinase.
We have these three blocks which can’t be reversed by the enzyme that moves it in the forward direction so here we need some special enzymes which can reverse the reaction to make glucose.
We are going to discuss these steps one by one.
Do you mean “Pyruvate to phosphoenolpyruvate”?
Exactly, because pyruvate kinase can’t reverse the process so it needs another special enzyme to reverse it.
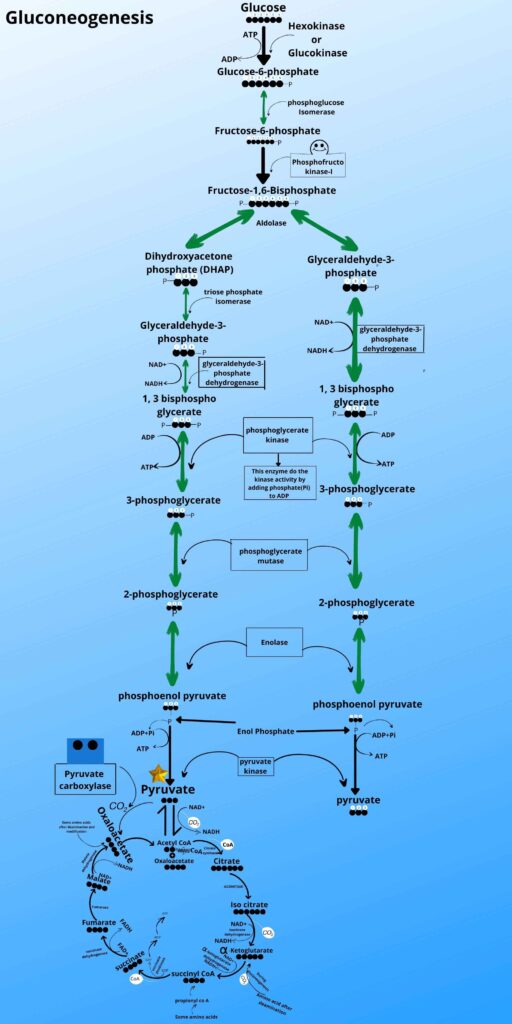
Do you see pyruvate in the Krebs cycle in the picture?
Yes, I can see it.
Here an enzyme called pyruvate carboxylase adds carbon dioxide (CO2) to pyruvate and converts it to Oxaloacetate.
How does pyruvate carboxylase work?
Manganese (Mn+), Magnesium (Mg+) ions, and Biotin vitamins are required for pyruvate carboxylase functioning while they require ATP for glucose production because we are going to make a molecule.
Biotin is very important for pyruvate carboxylase because it is the Biotin in pyruvate carboxylase which helps this enzyme to transfer the CO2 to pyruvate.
Here I want to tell you about a medical condition.
What is that situation?
Eggs especially uncooked eggs have a special protein called Avidin which binds avidly/strongly with biotin so for those people who eat uncooked eggs, their biotin is not available which then stops the functioning of pyruvate carboxylase and eventually lead to trouble in the gluconeogenesis pathway.
But how this enzyme is activated?
It is activated by acetyl Coenzyme A present in mitochondria for the Krebs cycle. As already discussed that after lipolysis glycerol and fatty acids are released, fatty acids move to mitochondria and are converted to Acetyl Coenzyme A which then activates pyruvate carboxylase and indirectly stimulates gluconeogenesis.
Also, the fatty acids after conversion to Acetyl Coenzyme A don’t activate the pyruvate carboxylase and reverse the Krebs cycle and make the glucose after reversing glycolysis too.
Now, what will be happened to oxaloacetate?
The pyruvate is converted to oxaloacetate but still, we have a problem.
What is it?
The problem is that the oxaloacetate is a very important component of the Krebs cycle so it can’t leave the mitochondria.
So what do cells do then?
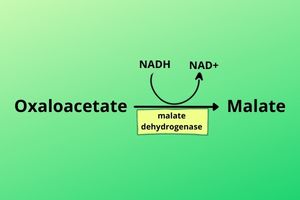
The cell breaks NADH to NAD+ and releases the hydrogen to cover the oxaloacetate by an enzyme called mitochondrial Malate dehydrogenase. The resultant compound after covering by hydrogen is now called Malate. Now the Malate can leave the mitochondria.
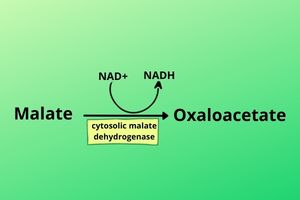
In the cytosol, the same malate dehydrogenase enzyme is present where it is called cytosolic malate dehydrogenase which converts back the Malate to oxaloacetate by removing hydrogen from it.
This process of malate coming out is called the Malate shuttle.
That’s nice.
Still, we didn’t get phosphoenolpyruvate?
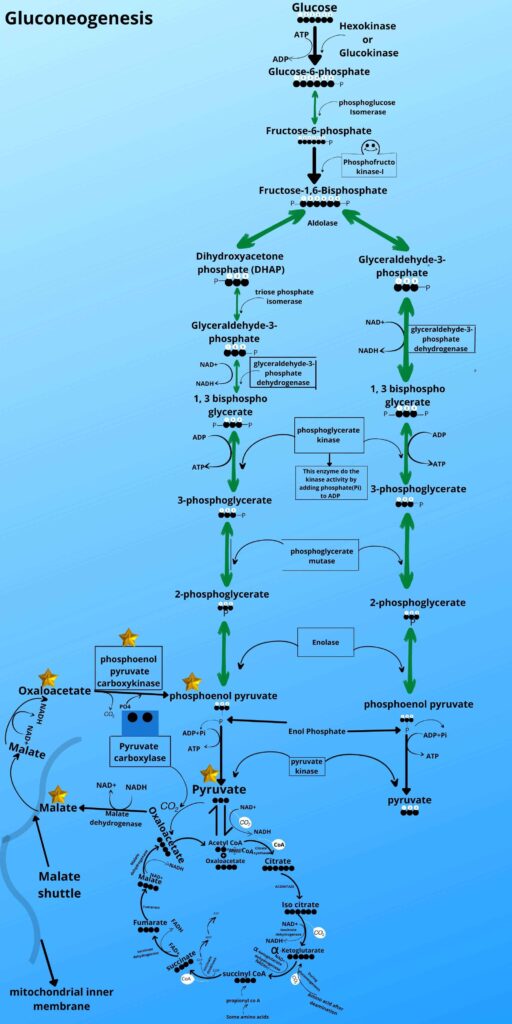
Don’t worry, here is the last step in which the oxaloacetate is converted to phosphoenolpyruvate by the phosphoenolpyruvate carboxykinase enzyme. This enzyme removes carbon dioxide as it was added before and adds phosphate to it to make phosphoenol pyruvate because the kinase enzyme adds phosphate to the molecule.
Now, what will happen?
As shown in the picture the phosphoenolpyruvate is then converted back to 2 phosphoglycerates, 3 phosphoglycerates, 1,3 bisphosphoglycerate, glyceraldehyde-3-phosphate, and Fructose 1, 6 bisphosphates respectively by the same enzyme as it was converted forward in glycolysis.
But here is another block point that can’t be directly reversed by phosphofructo kinase-I as it did forward direction.
Before going to reverse this step let me review their forward action first then you will understand the reverse step.
Do you know which enzyme converts Fructose-6-phosphate to Fructose 1,6 bisphosphate?
That was phosphofructo kinase-I.
That’s good.
But who activated this enzyme?‘
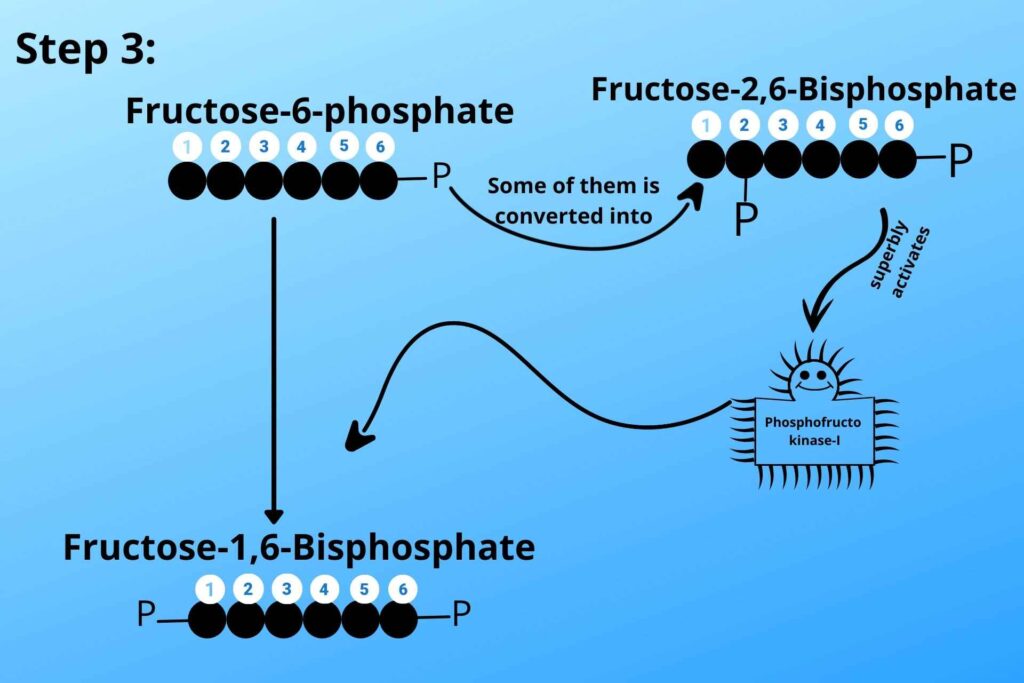
During the conversion of Fructose-6-phosphate to Fructose 1, 6 bisphosphates some of them are converted to Fructose 2, 6 bisphosphates by phosphofrcto kinase-II as it is adding phosphate at Carbon number 2. The concentration of Fructose 2, 6 bisphosphates also inhibits another enzyme called Fructose 1, 6 bisphosphatase which reverses the reaction by converting Fructose 1, 6 bisphosphate to Fructose-6-phosphate. The Fructose 2, 6 bisphosphate then strongly stimulates phosphofructo kinase-I to convert fructose-6-phosphate to Fructose 1, 6 bisphosphate by adding phosphate at carbon 1 and proceed the pathway in the forward direction.
But who activated phosphofrcto kinase-II?
There is a hermaphrodite enzyme that has two parts one is phosphofrcto kinase-II (kinases adds phosphate to the molecule) and the other part is called Fructose 2, 6 phosphatase as it removes phosphate from Fructose 2, 6 bisphosphate (phosphatase remove phosphate).
But which part of the enzyme (kinases or phosphatases) are activated, how do they decide?
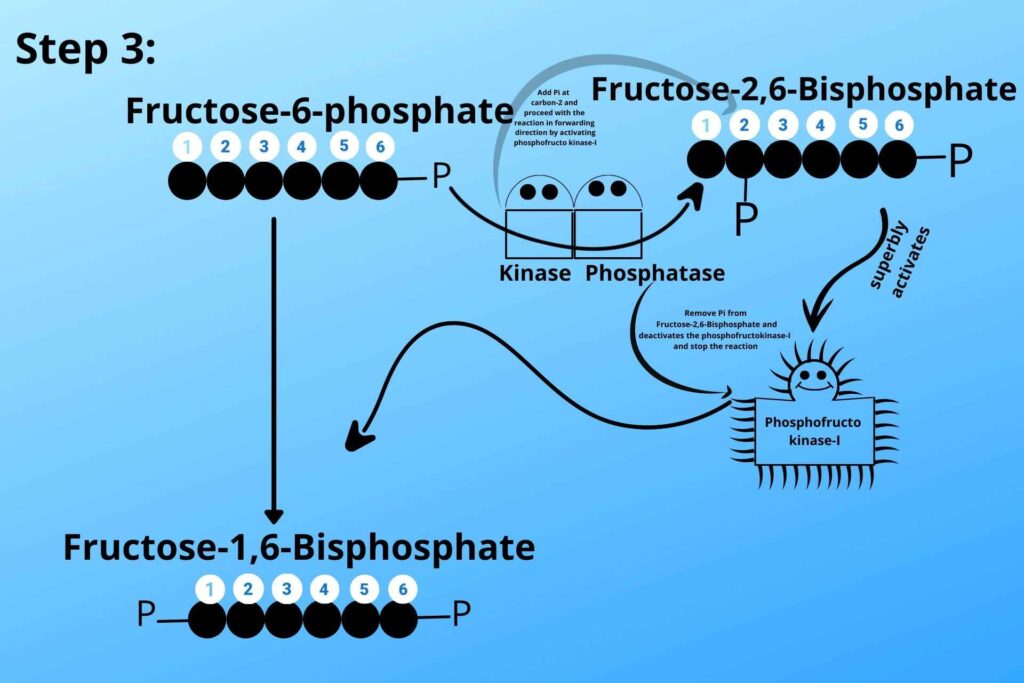
When Fructose-6-phosphate is high the phosphofrcto kinase-II is activated which converts some of Fructose-6-phosphate to Fructose 2, 6 bisphosphate which in turn activates phosphofrcto kinase-I and proceeds the reaction in the forward direction.
But how Fructose 2, 6 phosphatase is activated, how do cells know?
But when their less glucose in the body the Fructose 2, 6 phosphatase is activated which removes the phosphate from Fructose 2, 6 phosphatase which in turn stops the activation of phosphofrcto kinase-I and eventually stops the forward reaction.
How does it happen?
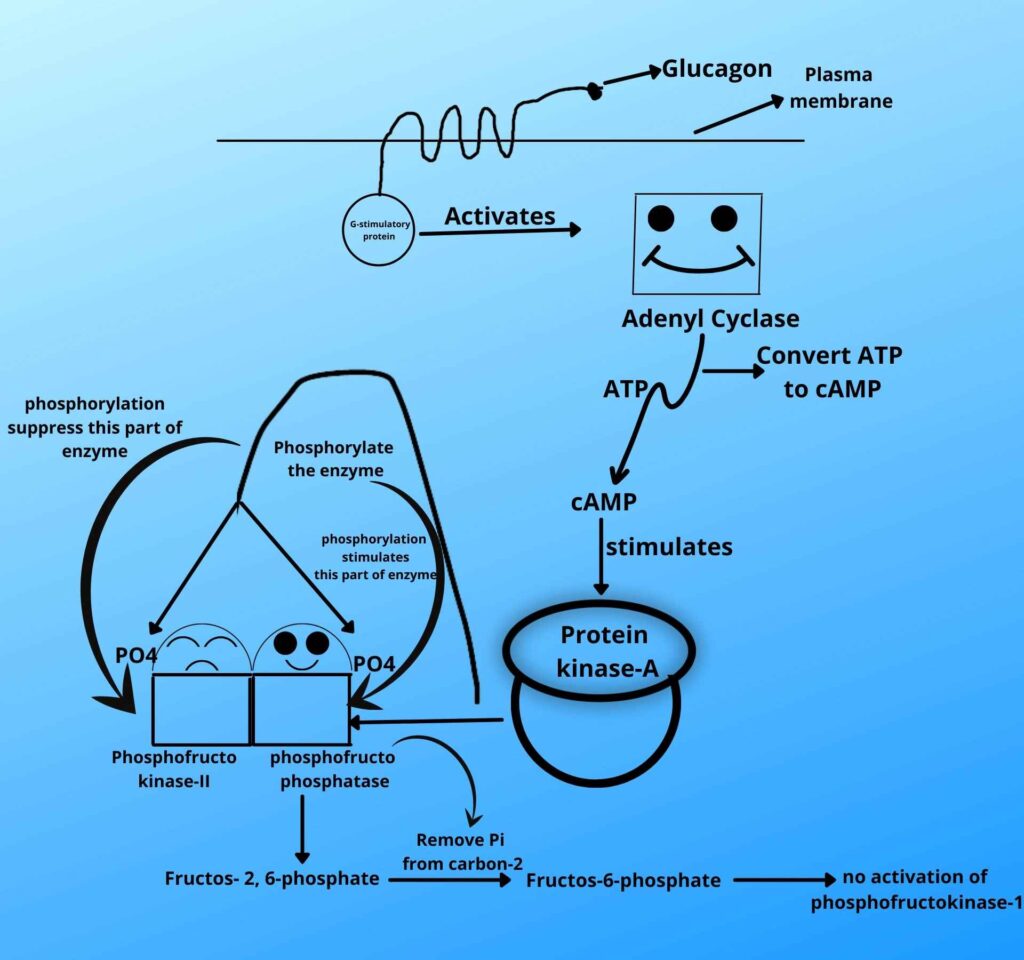
When the level of glucose is low glucagon is released which then binds with their receptor on the plasma membrane and this stimulated receptor is then coupled with G-stimulatory protein in the cell.
The G-stimulatory then stimulates adenyl cyclase which converts ATP to cAMP (cyclic AMP) and the number of cAMP in the cell increases. The cAMP in turn activates protein kinase A enzyme which phosphorylates both Fructose 2, 6 phosphatase, and phosphofrcto kinase-II enzyme.
In such a situation kinase activity is inhibited and phosphatase activity is activated so it means the Fructose 2, 6 phosphatase is activated which removes phosphate from Fructose 2, 6 bisphosphate, and when phosphate has removed the concentration of Fructose 2, 6 phosphate decreases which then stop stimulating phosphofrcto kinase-I. This will lead to the stoppage of the forward reaction.
As already discussed that high concentration of Fructose 2, 6 bisphosphate inhibit Fructose 1, 6 bisphosphatase enzyme which reverses the Fructose 1, 6 bisphosphate to Fructose-6-phosphate but now the concentration of Fructose 2, 6 bisphosphate decreases so it cannot keep 1, 6 bisphosphatase inhibited so it will be activated and convert back the Fructose 1, 6 bisphosphate to Fructose-6-phosphate so in this way this step is reversed.
Now let me tell you some of the roles of “protein kinase A”.
When glucagon level increases through multiple steps it activates protein kinase as discussed earlier it not only phosphorylates hermaphrodite (Bifunctional)enzyme but also phosphorylates pyruvate kinase which is inhibited and stops the glycolysis. This protein kinase also phosphorylates pyruvate carboxylase and phosphoenolpyruvate carboxykinase and activates them and reverses the steps for gluconeogenesis as discussed earlier.
This protein also phosphorylates certain transcriptional factor and this transcriptional factor go to the nucleus and will activate the gluconeogenesis pathway enzymes.
I think you are tired of repeating the names and maybe you get confused but read carefully and logically you will understand everything.
Let’s move to the next step. The Fructose-6-phosphate is converted back to Glucose-6-phosphate by the same enzyme i.e phosphoglucose isomerase who converted it into Fructose-6-phosphate in the forward direction of glycolysis because their enzyme is a two-way enzyme.
Now we are near to finish it by converting it to just glucose?
No, not. Again here is block because Glucose-6-phosphate can’t be reversed by the same enzyme i.e hexokinase who converted it in a forward direction because it is a one-way enzyme.
Do you know that cells have endoplasmic reticulum?
Yes, we know.
But what is the function of the endoplasmic reticulum here?
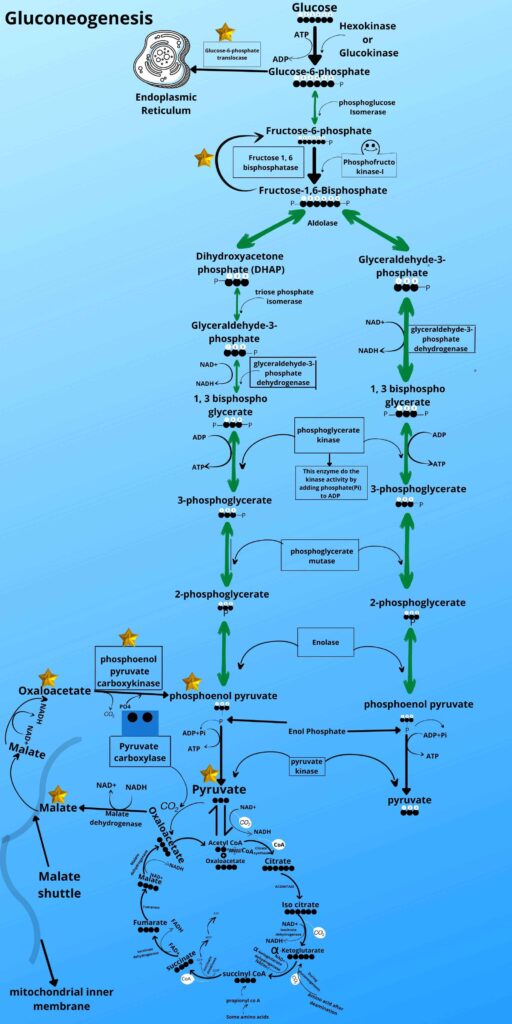
A very important function. The endoplasmic reticulum has a special transporter protein called Glucose-6-phosphate translocase which will take the Glucose-6-phosphate and transfer it to the Endoplasmic reticulum.
Okay.
Then?
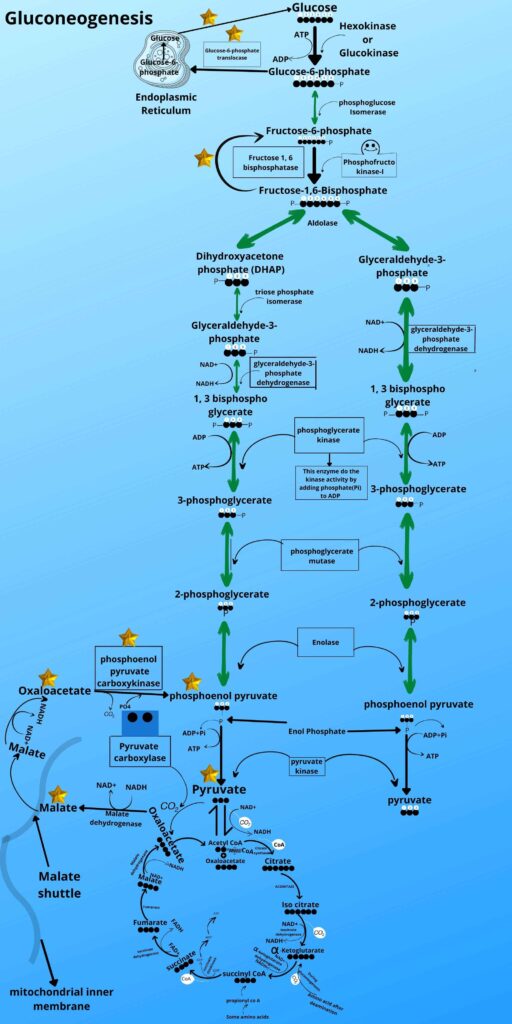
The endoplasmic reticulum membrane has an enzyme called Glucose-6-phosphatase. This enzyme has a functional domain in the lumen of the Endoplasmic reticulum where it converts the Glucose-6-phosphate to glucose by removing phosphate from it and as already mentioned many times that phosphatase removes phosphate from the molecule, so the product of gluconeogenesis (glucose) ends the gluconeogenesis process. The glucose will move from hepatocytes to blood and muscle back through the gluconeogenesis pathway.
This cycle of pyruvate and lactate to hepatocytes from muscle cells and then the formation of new glucose molecules from it and then returning of glucose to the muscle is called CORI cycle, besides fasting state this happens when muscles are doing constantly and heavy exercise.
As already discussed that gluconeogenesis occurs in the liver and kidney, do you know why?
No.
Because the Glucose-6-phosphatase enzyme is only present in liver and kidney cells that’s why gluconeogenesis occurs only in the liver and kidney.
Conclusion/summary
Again I would like to say that please first read and learn glycolysis and Krebs cycle first because 80% of your gluconeogenesis will be covered by learning glycolysis and Krebs cycle. In the gluconeogenic pathway, the three irreversible steps of glycolysis are reversed where three steps have problems because they are one way and can’t be reversed directly so we have to find the alternative pathway and raw materials for the reversal process. The purpose of gluconeogenesis is to make glucose when there is no or less glucose from non-carbohydrate sources. These all terms and steps of gluconeogenesis are explained so that you know why we need gluconeogenesis and when does gluconeogenesis occurs.






1 thought on “Gluconeogenesis pathway with enzymes: Introduction, Steps, Regulation, and Function”